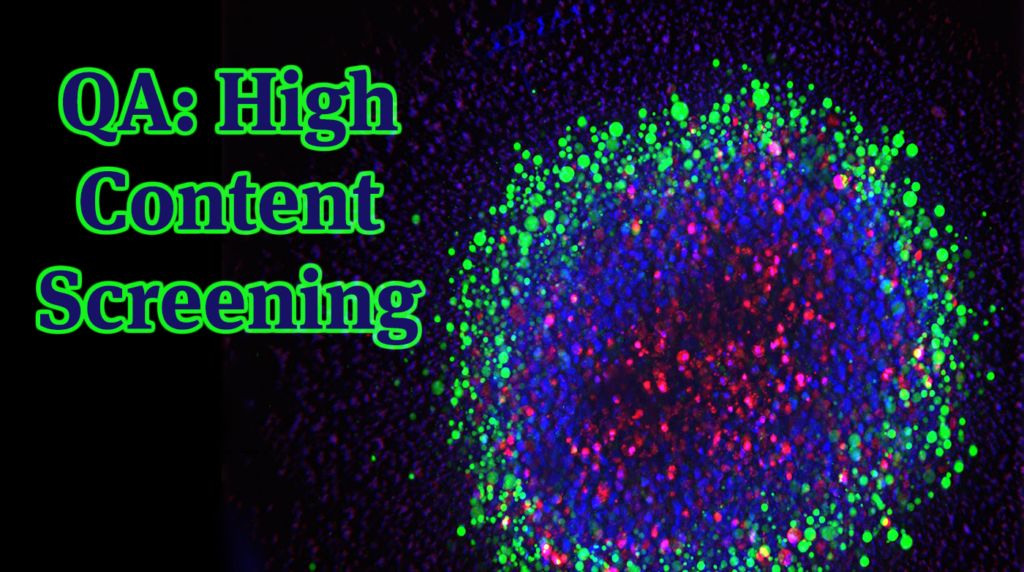
Within Imperial Brands’ Hamburg laboratory network is a Thermo CellInsight CX5 High Content Screening device.
But what does it do, and how is it helping our scientists to better understand our next generation products (NGP) and support our ambition to make a meaningful contribution to consumer health through tobacco harm reduction?
Science Communications Manager Rob Taylor spoke to Advancing Toxicology Manager Edgar Trelles Sticken to learn more about this important tool for in-vitro analysis.
Hi Edgar; what exactly is High Content Screening?
High Content Screening (HCS) is an advanced laboratory technology used to help identify changes in cellular phenotypes (i.e. visual or structural alterations) at the single‑cell level.
It combines fluorescent staining, automated microscopy, and software image analysis.
By simultaneously staining multiple protein targets (called epitopes) within each cell, HCS provides us with valuable insights into various cellular pathways activated by representative extracts generated either from our next generation products (NGP), or neat liquids/constituents – and allows us to compare the results against cigarette smoke.
As this approach captures numerous parameters across thousands of cells, it generates a very large amount of data per experiment – hence ‘High Content Screening’.
Why is it potentially an important part of the scientific assessment of our NGP?
As a responsible manufacturer dedicated to safeguarding our consumers, we believe High Content Screening adds an important new dimension to how we assess next‑generation products through our multi-stage, multi-discipline scientific assessment framework – and also as part of our innovative Alternatives to Animal Testing programme.
Traditional tests mainly search for genetic damage – the kind of changes that can lead to cancer. But smoking is linked to many other diseases, such as COPD and heart disease, which aren’t fully covered by those tests.
By focusing on individual cells, HCS technology can help us spot early signs of stress or inflammation – subtle changes that may be the first steps toward a range of smoking‑related conditions. In simple terms, it provides us with a broader picture of how our NGP interact with human biology, beyond just cancer risk – and is therefore a potentially important addition to current toxicological testing batteries.

Is HCS testing conducted in-house? What kind of facilities and accreditation do we need to perform the research?
HCS technology has been developed and carried out in-house (our Hamburg laboratory) since 2017.
As the projects conducted on our HCS platform are used to gain ‘add-on’ knowledge and are not performed on a routine basis, there’s no official accreditation on the horizon.
However, the device is integrated into an ISO 17025 laboratory environment which gives us confidence around the standardisation of the processes underpinning HCS testing – like cell culture best practice and rigorous staff training.
Briefly, what kind of results have we achieved focusing on substantiating the harm reduction potential of our various NGP compared to cigarettes?
We’ve already demonstrated that, following exposure to NGP extracts or neat liquids/constituents all endpoints measured (i.e. markers of cell health, such as stress, inflammation, and DNA damage) show no response – or responses only at significantly higher concentrations – compared to equivalent cigarette smoke extracts.
Our results to-date align with our relative risk scale comparing the harm reduction potential of NGP relative to cigarettes.
How are we building momentum around the importance of HCS externally?
Our results to-date have been published in the academic literature, and we also continue to present new data at CORESTA and other relevant conferences when the opportunities arise.
Does the laboratory collaborate with other teams, either internally or externally, to maximise the impact of our HCS data?
Internally, we’re supported by Imperial’s Data Science Lab team in France to efficiently handle the huge amounts of data generated during such analyses. We are very grateful for their assistance!
Externally, CORESTA’s next generation toxicity testing (NGTX) Task Force has endorsed HCS technology, while Interlab comparison studies have confirmed other platforms and laboratories deliver similar responses when using the same cellular model and positive controls for defined endpoints.
How do we ensure our data is reproducible and transparent enough to withstand regulatory scrutiny?
As mentioned earlier, internal reproducibility is checked through the use of positive controls. All our data are stored on dedicated servers, so we can provide raw data for audit purposes.
For external reproducibility, we’ve transparently detailed our methodology in published documents, so all qualified labs should be able to reproduce the data.

However, to reiterate HCS is still in the exploratory phase and therefore not currently part of our routine testing battery for NGP.
Are there any limitations to current HCS approaches?
Normally we are in favour of testing our NGP by direct exposure of our target cells with whole fresh aerosol (as published in Marina-Lacasta et al. 2025), or cigarette smoke.
This allows us to compare results on a per-puff base – a consumer relevant measure.
One limitation with HCS, as mentioned earlier, is that we’re currently limited to using aerosol extracts and/or neat substances/liquids.
However, in some cases suitable workarounds can be achieved.
How do you see HCS potentially evolving to meet increasing regulatory demands in the future?
Right now, regulators haven’t set official guidelines for High Content Screening. But that could change as the technology matures. One exciting area is the use of AI, is already helping us to handle and better interpret the huge datasets HCS produces.
In the meantime, industry groups like the CORESTA NGTX task force are working together to shape best‑practice recommendations – from standardised lab environments to strong cell culture principles.
This collaboration means that if regulatory bodies do move towards HCS in the future, the science will be ready to support for the benefit of consumer health.
Follow us on LinkedIn and read more about our intentions to help create potentially healthier futures for our consumers on our corporate website. You are free to share this content with credit to Imperial Brands under a Creative Commons Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license.
