As part of our commitment to making a meaningful contribution to tobacco harm reduction (THR), Imperial Brands continues to assess the THR potential of our Next Generation Product (NGP) portfolio relative to combustible tobacco.
In this latest study, published in the journal Applied In Vitro Toxicology, two of our commercially available nicotine pouch products were compared to combustible cigarette smoke across three established regulatory toxicological assays:
Under the conditions of test, the results were as follows:
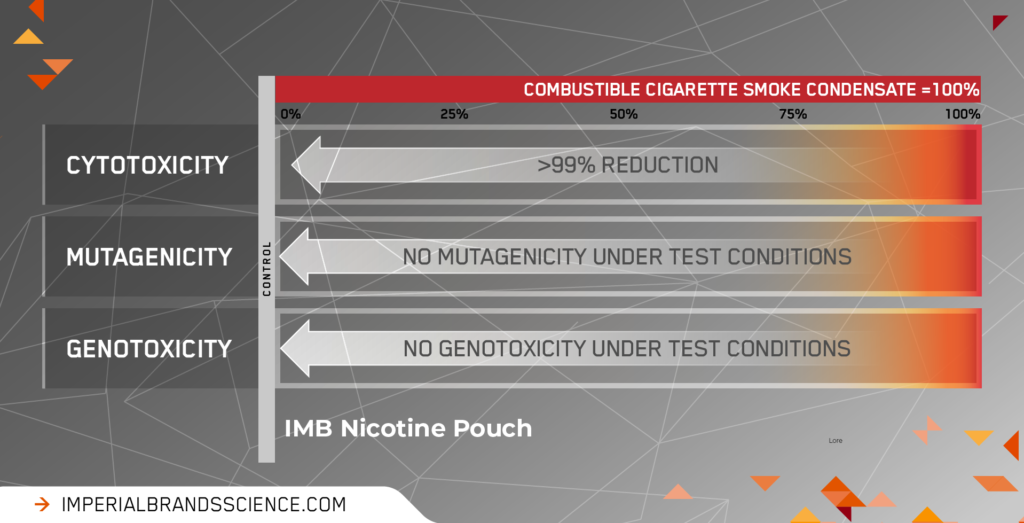
Figure 1: Representative average reductions in in vitro toxicity for nicotine pouches compared to combustible cigarette smoke
Matthew Stevenson, Investigative & Mechanistic Toxicity Senior Manager and study co-author, commented: “It’s encouraging to see the reduced levels of toxicants in our nicotine pouch products compared to combustible cigarette smoke translating directly into substantially reduced in vitro biological responses.
“This latest study was undertaken as part of wider research across our Scientific Assessment Framework as we seek to demonstrate to a range of stakeholders – including regulators, public health bodies and consumers – how these NGPs can play a positive role in THR strategies.
“We look forward to publishing further substantive research around these products in the near future.”
You are free to share this content with credit to Imperial Brands under a Creative Commons Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license.
