//Alternatives to animal testing
Posted 04/08/2021 9:19am
Imperial Brands doesn’t test its products on animals. Instead, our scientists use cutting edge in-vitro techniques. Launching a new section of our science website, Dr Liam Simms explains more…

Imperial Brands is committed to making a meaningful contribution to tobacco harm reduction (THR). From research to innovation, safety and compliance, the science of designing, engineering, manufacturing and marketing effective Next Generation Products (NGPs) for adult smokers is as challenging as it is rewarding.
Part of this commitment involves ensuring we continue to uphold our stated position not to commission or conduct research involving live animals (also known as in-vivo testing) for the scientific substantiation of our NGPs, unless formally required to do so by governments or by recognised regulatory authorities.
To this end, and our commitment to scientific transparency and openness, we’re pleased to launch a new webpage dedicated to our research around alternatives to animal testing. As our portfolio of NGPs progress through our Scientific Assessment Framework, a series of advanced and innovative in-vitro cell-based assays – many of which are informed by Toxicity Testing in the 21st Century (TT21C) methodologies – replace the need for any animal testing. (See our Q&A for further reading around TT21C.)
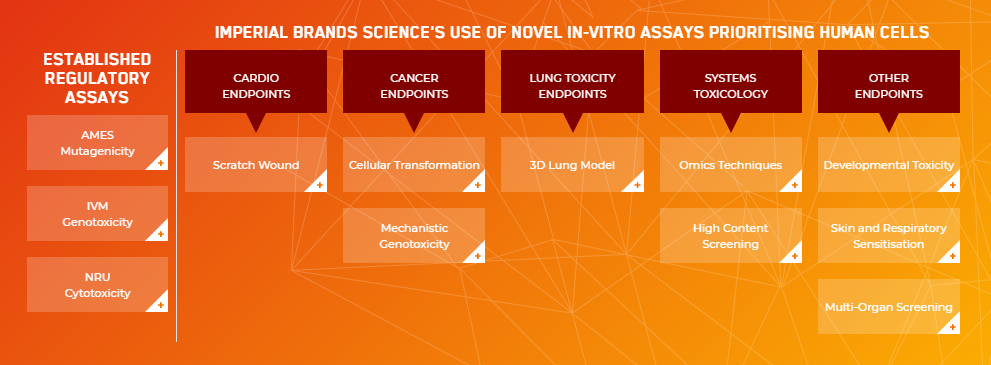
Our current TT21C assays – visit our new webpage to discover more…
The webpage also features an interactive table. It details the established regulatory assays our scientists use regularly to test for mutagenicity, genotoxicity and cytotoxicity, as well as explaining the wide selection of TT21C cellular assays we’re using to better understand the biological effects of our NGPs on various human endpoints. These include the scratch-wound assay for cardiovascular endpoints, 3D lung model for lung toxicity endpoints, plus our use of omics technologies and high-content screening. Additional functionality allows users to explore what papers and presentations our scientists have published around each assay to-date.
Endorsement of our approach 
Our alternatives to animal testing, TT21C-driven approach for the scientific assessment of NGPs has been well received externally. Our scientists have presented our in vitro research at scientific conferences across the globe, while our methodologies have also been recognised and endorsed by PETA Science, the Science Consortium of People for the Ethical Treatment of Animals.
Surprisingly, there’s been very little innovation in traditional in vivo methods over time; scientists are still conducting similar studies to those used almost 70 years ago. This is startling, considering animal testing isn’t a completely efficient or reliable tool for research, particularly for the assessment of NGPs, due to species differences and a lack of understanding about how observations seen in animals relate directly to humans.
Put simply, the best predictive model for a human is a human. We believe the capability of in vitro methods is so advanced when coupled with human clinical studies that animal in vivo methods for biological testing for the assessment of NGPs and their tobacco harm reduction potential have essentially been rendered redundant. For example, smoking-related diseases cannot be replicated in the animal models. And recent advances in alternatives to animal testing now allow for a reduction in the many animals used in routine testing.
 These techniques have incredible scientific potential, but TT21C remains a novel framework for NGP assessment in several aspects – not least because it’s just over a decade old. This means there’s still lots of work to do in not just continuing to develop the assays and advance in vitro science, but also around educating regulators, media and consumers around its advantages.
These techniques have incredible scientific potential, but TT21C remains a novel framework for NGP assessment in several aspects – not least because it’s just over a decade old. This means there’s still lots of work to do in not just continuing to develop the assays and advance in vitro science, but also around educating regulators, media and consumers around its advantages.
We firmly believe the TT21C-inspired alternatives to animal testing blueprint is a toxicological game-changer, and our latest NGP research is delivering the results to prove it.
We hope you enjoy our new webpage.
—

You are free to share this content with credit to Imperial Brands under a Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license.
