This article was updated on 29/02/24 to reflect additional findings within our academic literature review exploring the potential contribution of tobacco-free oral nicotine pouches to tobacco harm reduction. The open-access paper has been published in the Cureus Journal of Medical Science. You can also find a summary of our own science to-date on our product, zoneX, here.
Tobacco harm reduction (THR) represents one of the most promising global public health policies with the potential to save many lives if fully embraced.
While science suggests nicotine is an addictive substance and not risk-free, numerous public health bodies worldwide – including the Royal College of Physicians, the US Food and Drug Administration and the Public Health England to name a few − recognise that it isn’t the primary cause of smoking-related diseases. Likewise, the scientific consensus to date also suggests nicotine does not, by itself, cause cardiovascular disease.[efn_note]Royal College of Physicians (2016) Nicotine Without Smoke , Center for Disease Control (2014) Surgeon General’s Report: The Health Consequences of Smoking—50 Years of Progress [/efn_note]
Importantly, nicotine can be delivered through products along a scale of risk.
Free from harmful tobacco combustion, high quality nicotine pouches offer adult smokers who are unwilling or uninterested to stop smoking a potentially harm-reduced alternative. Its appearance and oral method of use is similar to traditional tobacco-containing Scandinavian snus, itself a harm-reduced product supported by favourable prevalence data in Sweden (see our snus infographic here for more information). Critically, one vital difference makes modern nicotine pouches very distinct: they are completely free of tobacco leaf. As such, their THR potential relative to cigarettes and snus may be even greater.
The case for nicotine pouches

These small, pre-formed pouches contain high-purity pharmaceutical grade nicotine either sprayed onto or a plant fibre-based substrate, such as wheat or bamboo fibres. They also contain high-quality flavourings. humectants to prevent drying out, and other additives to ensure product stability.
By placing a pouch under the lip for up to an hour, nicotine is released into the bloodstream via the gum lining (oral mucosa).
Nicotine pouches are likely to be significantly less harmful than continued cigarette smoking because they:
These factors mean nicotine pouches sit furthest away from cigarettes on our relative risk scale.
Fewer and lower levels of toxicants
All next generation products (or NGP, including vapes and heated tobacco) provide a potentially less harmful alternative to cigarettes because they contain far fewer and substantially lower levels of the toxicants generated by burning tobacco.
Our research shows levels of notable toxicants of public health interest in our own high-quality nicotine pouches are substantially lower compared to tobacco smoke – if they’re even present in quantifiable levels at all.
Of the 16 toxicants assessed in Imperial Brands’ nicotine pouches, only four were found in measurable levels: acetaldehyde, formaldehyde, nickel and chromium. Compared to cigarette smoke, levels of acetaldehyde and formaldehyde were reduced by >94%. For nickel and chromium the very small amounts observed were below the regulatory limits set for medicinal products.
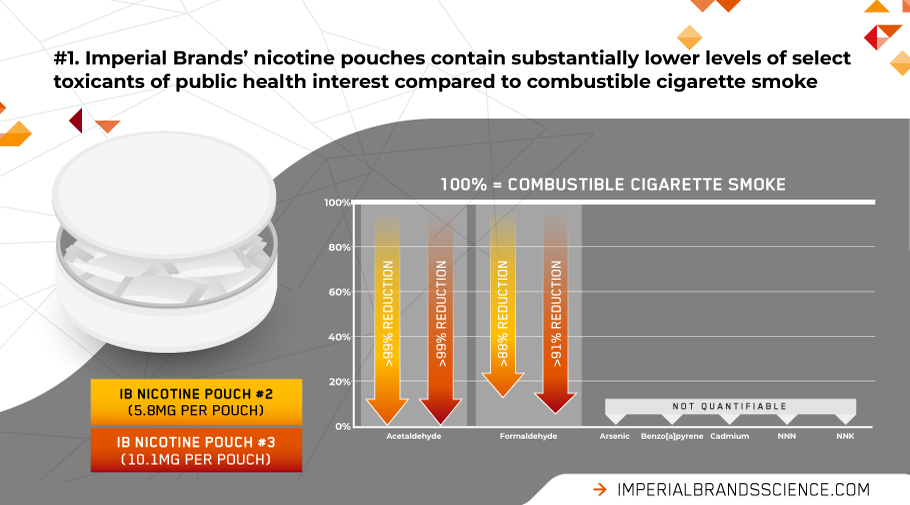
Fewer toxicants translates to less toxicity to cells
Studies on other high quality nicotine pouches have observed similar reductions with very low, or undetectable, levels of several select toxicants of public health interest commonly found in tobacco product comparators. [efn_note] https://sciences.altria.com/library/-/media/Project/Altria/Sciences/library/conferences/2020/SRNT-2020-Poster-Wagner-on-HPHCs.pdf[/efn_note]
Our lab studies also demonstrate the fewer and substantially lower levels of toxicants present in our nicotine pouches directly results in considerably reduced in vitro toxicity compared to cigarette smoke.
Across a battery of standardised and recognised regulatory assays, Imperial Brands’ nicotine pouches exhibit significantly reduced in vitro cytotoxicity, mutagenicity and genotoxicity responses.
Cytotoxicity kills and inhibits the growth of cells and is considered a potential step in several processes that lead to smoking-related disease. Mutagenicity measures of the ability of a substance to cause changes in the genetic material of a cell, while genotoxicity assesses damage to the structure or function of genetic material and has been linked with cancer development.
Read together these toxicities can indicate potential problems in a biological system, which is why we test for them as part of the Biological component in our Scientific Assessment Framework.
Imperial Brands’ nicotine pouches elicited a >99% reduction in cytotoxicity, more than 100 times lower than cigarette smoke condensate, and produced no mutagenicity or genotoxicity under conditions of the test, with results comparable to the negative control. [efn_note] Fold reduction of 618 for BEAS-2b and 161 for HepG2 for our nicotine pouch product #3 (8.96mg), when normalised to nicotine content.[/efn_note]

Our in vitro testing in other areas relevant to our robust product stewardship standards also indicates our nicotine pouches don’t have skin sensitisation properties. In lab models assessing the gene expression profile of 200 markers in human cells lines, our nicotine pouches were not predicted to cause an allergic reaction or skin rashes.

In further in vitro studies, other researchers have assessed cell viability and cell health markers, as well as mechanistic genotoxicity and oxidative stress of nicotine pouches (ZYN) and tobacco product comparators.[efn_note] Bishop E (et al.) 2020. An approach for the extract generation and toxicological assessment of tobacco-free ‘modern’ oral nicotine pouches. Food and Chemical Toxicology, 145, p.111713. [/efn_note] This paper found:
Nicotine delivery
Satisfying nicotine delivery is integral to any NGP’s efficacy. To achieve their maximum THR potential, it is critical these products offer adult smokers a fulfilling alternative to cigarettes, encouraging a permanent transition away from combustible sources of nicotine.
Effective nicotine delivery is thus key to adult smoker acceptance.
Emerging clinical evidence demonstrates the efficient absorption of nicotine from the oral mucosa into the blood circulation from nicotine pouches, with no significant adverse effects associated with use under study conditions. [efn_note]Lunell, E., Fagerström, K., Hughes, J., & Pendrill, R. (2020). Pharmacokinetic Comparison of a Novel Non-tobacco-Based Nicotine Pouch (ZYN) With Conventional, Tobacco-Based Swedish Snus and American Moist Snuff. Nicotine & Tobacco Research. doi: 10.1093/ntr/ntaa068[/efn_note]
To understand how adult smokers’ bodies absorb nicotine from nicotine pouches, how long it stays in their body, and how this compares to the blood nicotine delivery of smoking, we conducted a single use pharmacokinetic (PK) study in a controlled clinical setting with 24 adult users of both oral nicotine products and cigarettes.
Our clinical data shows our nicotine pouches efficiently deliver nicotine to the blood of adult smokers via the oral mucosa, and do not exceed the delivery profile of cigarettes.
This is evidenced in the blood nicotine delivery curve below that unequivocally shows nicotine is absorbed via buccal absorption, with limited (if any) swallowed. Swallowing would generate a flatter and lower shaped curve, with limited nicotine delivery to the blood owing to first-pass elimination coupled with low stomach pH. Furthermore, no late secondary nicotine peaks – indicative of the slower gastrointestinal absorption of nicotine – were observed in the plasma profile following use of our products.
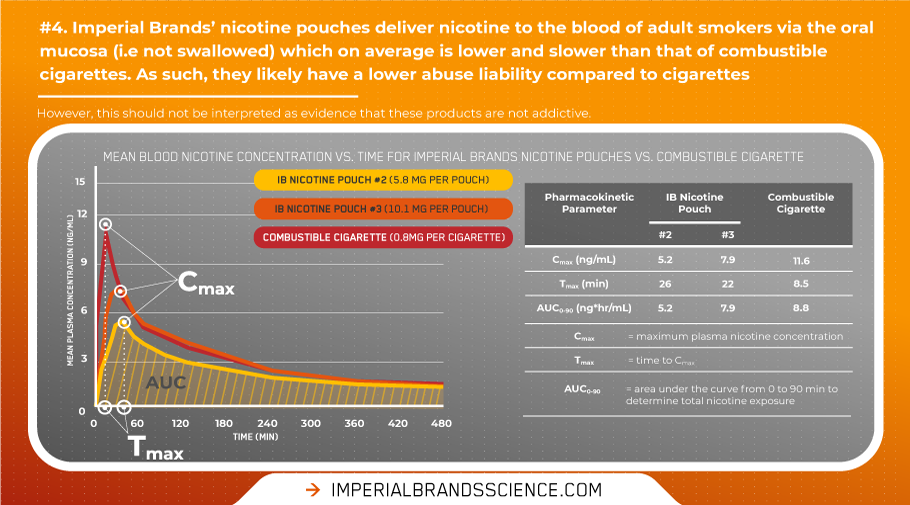
When compared on a per pouch/per cigarette stick basis, our nicotine pouches have a maximum nicotine concentration (Cmax) lower than that of cigarettes. This is coupled with a longer time to reach maximum concentration (Tmax), which – again − is consistent with oral rather than gastrointestinal nicotine absorption. The extent of nicotine exposure (AUC) is also substantially less compared to typical cigarette smoking indicating a lower total nicotine delivery over time.
As the blood nicotine delivery from our nicotine pouches is on average is lower and slower compared to cigarettes, these products are likely to have a reduced dependency potential relative to cigarettes. While this shouldn’t be interpreted as evidence that these products are not addictive, the data indicate the relative addictive potential of our nicotine pouches is unlikely to be greater than cigarettes among current adult smokers.
Adult smoker satisfaction
Effective nicotine delivery, as previously stated, is central to achieving THR for adult smokers. It’s therefore pivotal we investigate all its clinical aspects.
Examining blood delivery analyses just one branch of pharmacology. It’s also essential to understand how adult smokers feel after having used nicotine pouches (known as pharmacodynamics, or PD); in particular, their desire to smoke cigarettes again and − centrally to realising THR − whether these products are likely to be an acceptable alternative for adult smokers.
Following initial use of Imperial Brands’ nicotine pouches, our research shows significant reductions in desire to smoke cigarettes compared to the baseline starting point (preceding any nicotine product use). Nicotine pouches offered sustained reductions in desire to smoke for up to four hours before returning to the starting level. In contrast, for cigarettes, desire to smoke again returned to baseline much sooner (i.e. within less than an hour).
Across the usage period, our data suggest our nicotine pouches may also provide greater levels of satisfaction than a cigarette, indicating they are likely an acceptable alternative for adult smokers.
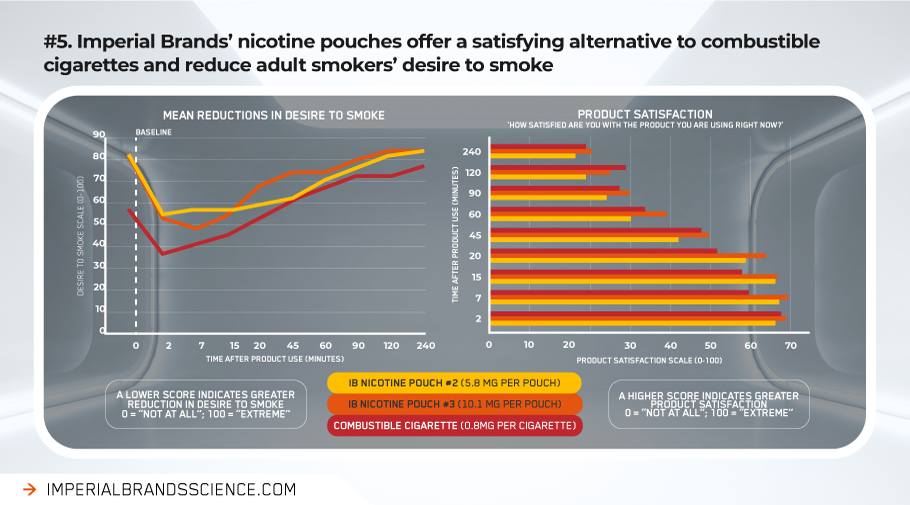
Other research from across the category reports similar encouraging results, with positive scores for nicotine pouches reduction in tobacco and nicotine withdrawal, as well as for satisfaction and pleasantness.[efn_note]https://sciences.altria.com/-/media/Project/Altria/Sciences/presentations/2020/SRNT-2020-Poster-Rensch-Characterization.pdf[/efn_note]
A promising short-term safety and tolerability profile
Data from our clinical study discussed above shows our nicotine pouches also have a favourable short-term safety and tolerability profile, with no serious adverse effects reported throughout the observation period.
Only one mild adverse event was observed: a case of hiccups considered likely related with use of the product. Hiccups are a known common, temporary and self-resolving event frequently reported with use of nicotine products.
Crucially, no clinically significant changes in laboratory safety samples, physical examinations and cardiovascular measures were observed.
Other published clinical research on nicotine pouches reports similar findings, with no serious adverse effects reported during the observation period.[efn_note]Lunell, E., Fagerström, K., Hughes, J., & Pendrill, R. (2020). Pharmacokinetic Comparison of a Novel Non-tobacco-Based Nicotine Pouch (ZYN) With Conventional, Tobacco-Based Swedish Snus and American Moist Snuff. Nicotine & Tobacco Research. doi: 10.1093/ntr/ntaa068[/efn_note] Although 16 minor adverse events were recorded, just two (dry mouth) were judged to have “possible or probable relationship to treatment [i.e. ZYN nicotine pouch product use].”

Biomarkers of Exposure
Biological markers are measurable in the body and allow scientists to quantify the extent of user exposure to smoking-related chemicals. The US regulator for tobacco and nicotine products, the FDA, has identified a set of 93 harmful and potentially harmful constituents in cigarette smoke, also known as HPHCs.
In clinical studies, scientists often ask subjects to switch from smoking to a NGP and then look at whether these levels of exposure to HPHCs are reduced. While Imperial hasn’t yet conducted our own BoE study focusing on the tobacco free oral nicotine pouch category, several external studies have have either assessed the potential impact of nicotine pouch use in humans; in some instances using biomarkers of exposure (BoE, indicators of exposure to harmful toxicants) and biomarkers of potential harm (BoPH, indicators of disease risk). Other related studies have either assessed disease/human health endpoints, such as damage to the oral mucosa or autonomic nervous system (ANS) dysfunction, or modelled the impact of nicotine pouches on all-cause mortality relative to smoking.
While the relatively low number of studies in this area suggests further examination of the potential impacts of nicotine pouches on various health endpoints at the population level is warranted, the research to-date suggests favourable changes in biomarkers among smokers who switch completely to using nicotine pouches and, therefore, their potential for THR.
Read about the findings in more detail in our academic literature review here.
Adult smokers motivation and intention to use
Finally, it’s important to consider behavioural data indicating nicotine pouch perceived usage and risk, as well as appeal for both intended use by adult smokers and potential unintended use by youth or non-smokers.
Consumer panel data from other studies indicates adult tobacco users find nicotine pouches moderately to extremely appealing, and are by far the largest group of regular users, with harm reduction and health reasons cited as the main reason for use.[efn_note]Plurphanswat N. et al. (2020). Initial Information on a Novel Nicotine Product. Am J Addict. 2020 Jul;29(4):279-286. doi: 10.1111/ajad.13020. Epub 2020 Mar 16. PMID: 32176374.[/efn_note]
Other research also shows positive transition rates for adult tobacco users moving to nicotine pouches: in one conference poster presentation, 28% of adult smokers and 72% oral tobacco users fully transitioned to nicotine pouches after only six weeks, suggesting high levels of adult tobacco user satisfaction and acceptance over potentially more harmful products.[efn_note]Presented from On! Altria GTNF Stand data[/efn_note]
Our own research likewise reveals similar encouraging results.
We asked 1532 adult tobacco and nicotine product users and non-users in the UK about their perceptions toward our nicotine pouches.
Encouragingly, of the current adult smokers who reported they would be likely to purchase nicotine pouches in order to try them, almost two-thirds said they would use them to replace some of or all their cigarettes.
THR reasons appear to be driving those motivations to try or purchase our nicotine pouches among adult smokers: “It might be less harmful to me than cigarettes” was by far the most popular choice. Recognising that nicotine pouches may aid transition away smoking, and have a much lower impact on bystanders compared cigarette smoke, featured as others.
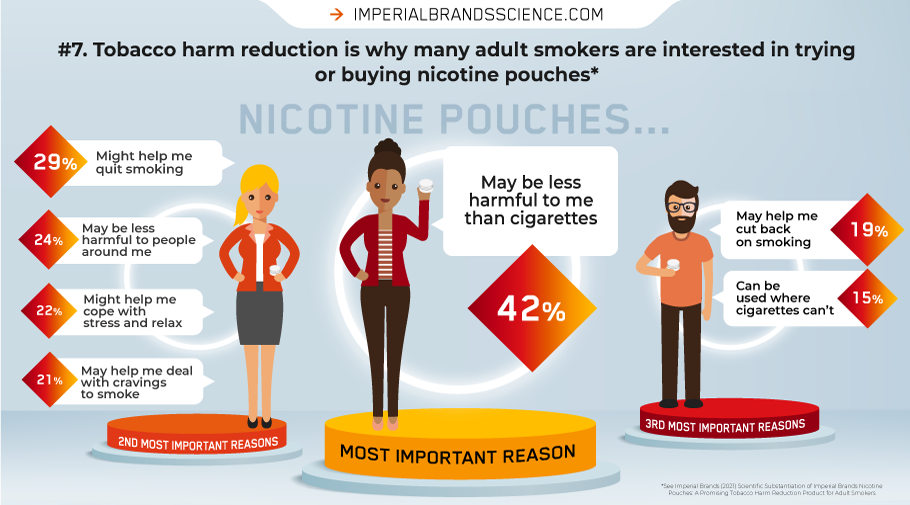
Furthermore, nicotine pouches were also perceived as likely having a lower risk than continued cigarette smoking, as well as presenting a lower perceived risk for developing lung cancer, mouth cancer and cardiovascular disease.
Critically, however, current former and non-smokers of tobacco and nicotine products all correctly understood that nicotine pouches contained nicotine, an addictive chemical, and were not risk-free.

Acceptable to current adult smokers but not appealing to non-smokers
Although these results are positive, it’s vital nicotine pouches aren’t used by vulnerable and non-smoking populations, and particularly by youth, because this will reduce their THR potential at the population level.
Data from our 1532 adult tobacco and nicotine product users and non-users UK survey reveal adult smokers are significantly more likely to try or purchase our nicotine pouches than both younger and older never-users, as well as long-term quitters.
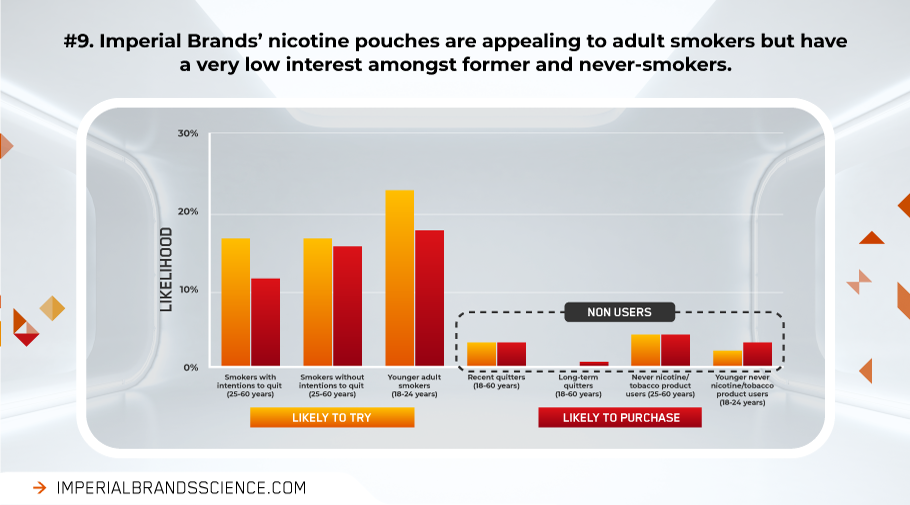
Curiosity and possible intent to initiate and purchase our nicotine pouches appears very low among never-smokers. As such, there is little evidence these products would serve as a ‘gateway’ to regular nicotine use or cigarette smoking in nicotine naïve populations.
Our findings align with other research indicating tobacco-naïve users and young adults have very little intention to buy, use or even try nicotine pouches, even following exposure to promotion and advertising. [efn_note]https://sciences.altria.com/sitecore/-/media/Project/Altria/Sciences/presentations/2020/SRNT-2020-Poster-McCaffrey-Evalution-of-Tobacco-Users.pdf[/efn_note] [efn_note]https://www.coresta.org/sites/default/files/abstracts/2020_ST16_Sarkar.pdf[/efn_note] Consumer panel data indicates adult tobacco users find nicotine pouches (ZYN) moderately to extremely appealing, and are by far the largest group of regular users. This study found 90% of non and former users did not find nicotine pouches appealing, only 3% of non-users and 2% of former users were interested in buying the product. [efn_note]Plurphanswat N. et al. (2020). Initial Information on a Novel Nicotine Product. Am J Addict. 2020 Jul;29(4):279-286. doi: 10.1111/ajad.13020. Epub 2020 Mar 16. PMID: 32176374.[/efn_note], [efn_note]Sudhanshu Patwardhan, Karl Fagerström. (2021). Nicotine pouches- a research and regulatory policy agenda to maximise public health benefits and minimise harms. Qeios. doi:10.32388/L4TIAF.2.[/efn_note]
Across the category, additional research in this area is clearly required. However, our initial findings add to the growing scientific evidence base that nicotine pouches are acceptable for current adult smokers while not being appealing to non-smokers. Furthermore, while nicotine pouches are widely understood as not risk-free, among adult smokers – who stand to benefit the most from transitioning to such products – they are perceived positively as a potentially less harmful alternative to cigarettes.
Unlocking the full public health potential
Our research complements the encouraging broader scientific literature to date and suggests high quality nicotine pouches:
So far, the sum of available scientific evidence – over 100 academic papers up to 31 May 2023 as per our review of the literature – indicates high quality nicotine pouches are likely to be substantially less harmful than continued cigarette smoking, and could play an important role in THR for adult smokers seeking alternatives.
Further scientific research, particularly more long-term clinical and behavioural studies, are needed and research in this category is ongoing.
In the meantime, further public health advocacy and scientific exploration is clearly warranted.
The concept of THR is simple and compelling: to encourage adult smokers who would otherwise continue to smoke to transition to less harmful products that deliver nicotine, but with fewer and substantially lower levels of toxicants associated with burning tobacco.
Adult smokers are more likely to transition to less harmful products if the product provides a satisfying experience, especially regarding nicotine delivery. This is why continued product innovations in the context of a responsible marketing and evidence-based regulation are crucial to making harm reduction a reality, thereby benefiting public health.
In countries – such as the UK, US, Canada and New Zealand – where reduced harm products have broadly been advocated by regulators and public health bodies they have already had a positive public health impact, and in some cases, contributed to significantly accelerated declines in smoking rates.
But as one of the most recent innovations in the NGP sphere, tobacco-free nicotine pouches have yet to make their mark.
We encourage governments and regulators to consider the promising body of scientific evidence. Not only should nicotine pouches be regulated according to their risk relative to cigarettes, in parallel they must be publicly and actively endorsed as a satisfying and potentially less harmful alternatives for adult smokers who would otherwise have continued to smoke.
Doing so will expediate and cement their contribution to THR.
—
You are free to share this content with credit to Imperial Brands under a Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license.
